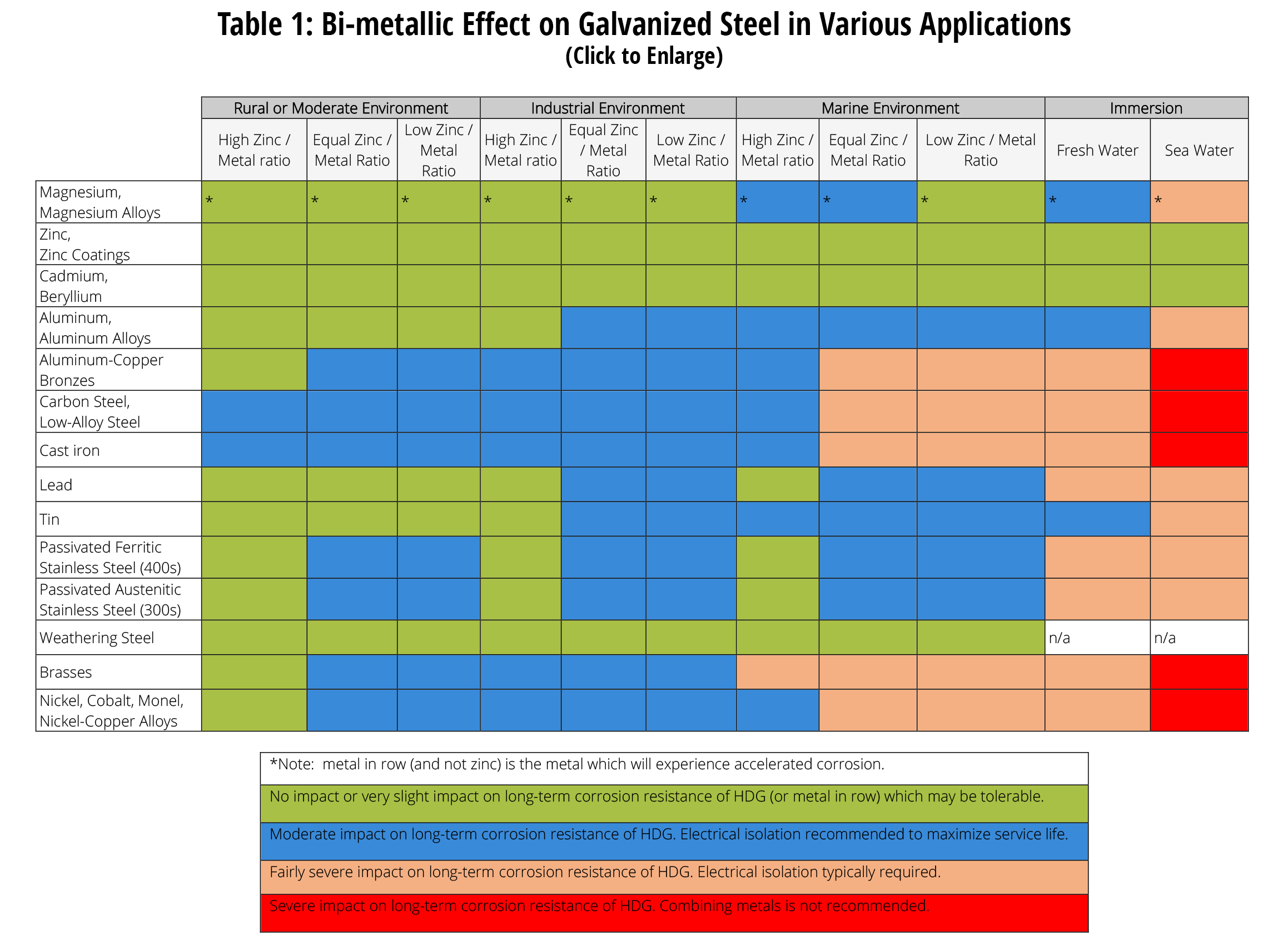
When two different metals are in contact in a corrosive environment one of the metals experiences accelerated galvanic corrosion while the other metal remains.
Galvanized steel sheet electrolysis.
Galvanized steel also can be coated with nickel copper etc.
The galvanized steel anodes should not be used in electrolysis.
If you break the conductive metal circuit between the copper and galvanized pipe galvanic corrosion electrolysis does not occur.
Stainless steel and galvanized materials often are found together in the industry with applications such as galvanized fasteners stainless steel pressure vessels and roof and siding panels.
Galvanic corrosion also called bimetallic corrosion or dissimilar metal corrosion is an electrochemical process in which one metal corrodes preferentially when it is in electrical contact with another in the presence of an electrolyte.
The presence of two dissimilar metals in an assembly is not always a sign of trouble but it could be a problem.
Hot dip galvanized steel is well suited for use in a variety of environments and fabrications and sometimes is placed in contact with different metals including among others stainless steel aluminum copper and weathering steel.
If zinc and or other coat metals get into the electrolyte they will form compounds which in their turn will contribute alloyed metals to the cathode some plating of the iron object being de rusted may occur.